Clinical, Regulatory, and
Quality for Emerging Biotech
Standardized, pre-configured Vault applications without the implementation and maintenance costs.
Announced 2024 | Status Early | Customers 1-10
Vault Quality
QualityDocs and Training
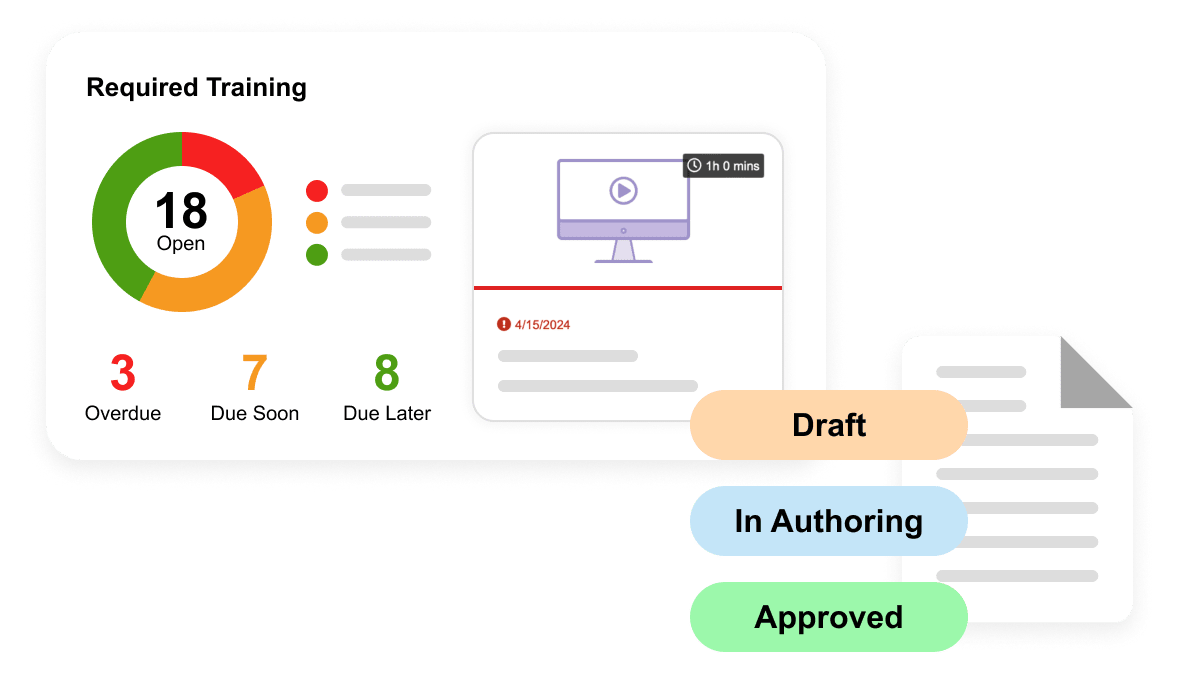
QualityDocs Basics is the industry-leading regulated quality content management solution. Including best practices for managing SOPs and other regulated content.
Vault Training Basics is an industry-specific LMS ensuring that your team is ready to perform their jobs while enabling GxP compliance and audit readiness.
Vault RIM
Submissions and Submissions Archive
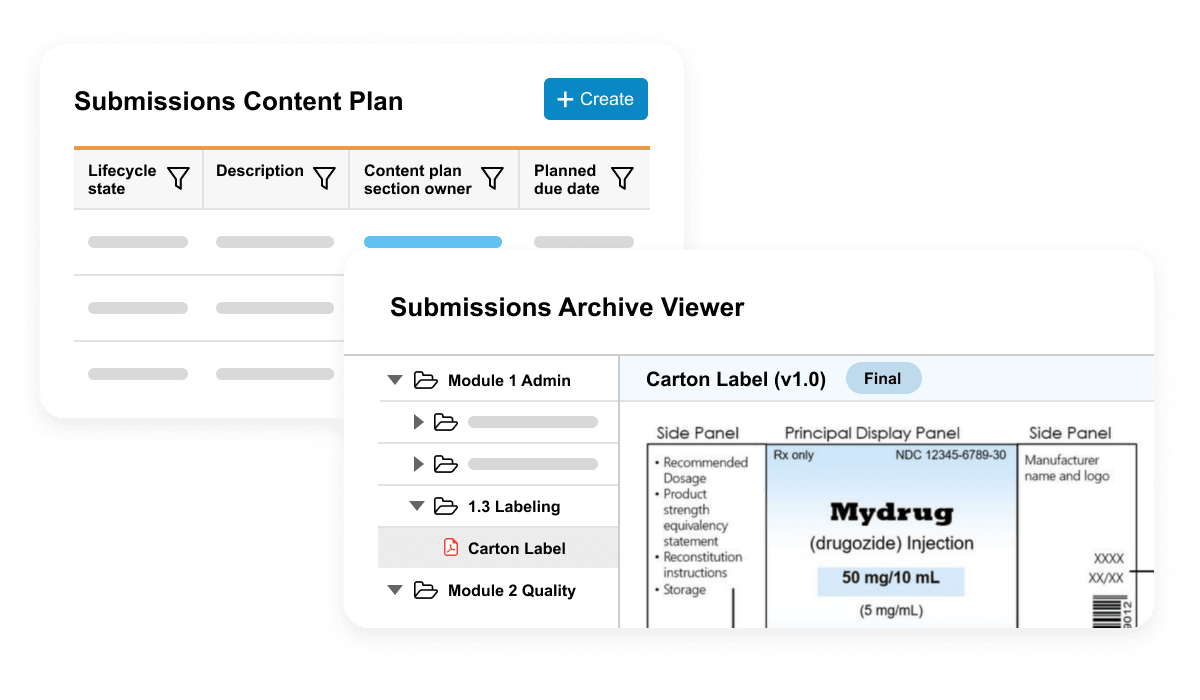
Vault Submissions Basics is used to plan, collaboratively author, review, and approve regulatory documents. It includes dashboards and reports to allow submission managers to track the status of documents in real time.
Vault Submissions Archive Basics is a secure repository of submission-published output and includes a viewer for easy access to historical applications submitted to health authorities.
Vault Clinical
eTMF

Vault eTMF Basics is the leading trial master file application used to ensure quality, timeliness, and completeness of a TMF. It provides full content management capabilities for upload, version control, QC/approval, and real-time co-authoring for study documents.
Vault Basics
Industry-leading Veeva Vault with zero implementation
Best of breed Vault applications now pre-configured and pre-validated to be up and running quickly and easily.
Global Support & Training
End-user 24x7 support provided by the Veeva Global Support Center. Robust on-demand training for each application available just a click away.
Pre-Validated
Access complete validation packages on-demand, with every release pre-validated by Veeva. Conduct self-service vendor audits instantly.
Always Current
Veeva-managed updates, delivered three times a year, keep your Vault current and ensure you benefit from the latest features.
Powered by Veeva Vault
Built for Emerging Biotech, Managed
and Maintained by Veeva









Learn more
Veeva Vault Basics
Let us know if you are interested in learning more about Veeva Vault Basics.

